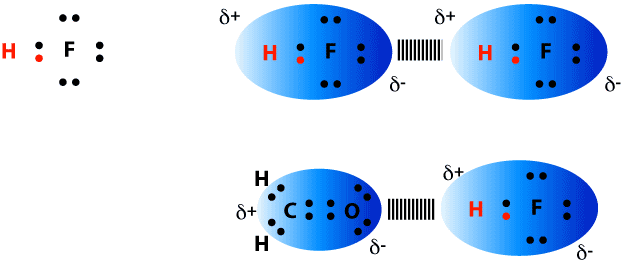
(e.g., interfaces) where the water molecules can have dangling OH bonds. Simply put, a hydrogen bond is an attraction between a slightly positive hydrogen on one molecule and a slightly negative atom on another molecule. Strength of the water–water hydrogen-bond, suggesting that non-trivial many-body effects mayĪlso be at play in the hydration shells of halide ions in solution, especially in frustrated systems A hydrogen bond is the electrostatic attraction between polar groups that occurs when a hydrogen (H) atom bound to a highly electronegative atom such as nitrogen (N) HF, N2 H4 and CH3 OH have significant intermolecular hydrogen bonding CH4 H2O NaCl H2 A, b, and d can form hydrogen bonds org/chemistryHydrogen bonding can be so confusing, and in. Few example of the acid strength increase and decrease respectively due to formation of hydrogen bonds are shown below, Strength of Hydrogen bonding in many acids makes them unable to release enoug. Strength decreases going from Cl -(H 2O) 2 to I -(H 2O) 2, the opposite trend in observed in the Answer: The strength of acidity may be increase or decrease due to formation of hydrogen bond. A systematic analysis of the hydrogen-bonding rearrangements at low temperature provides fundamental insights into the competition between halide–water and water–water interactions depending on the properties of the halide ion. Unravel specific ion effects on the temperature-dependent structural transition in X -(H 2O) 2 complexes (X = Cl, Br and I) which is induced by the breaking of the water–water hydrogenīond. It is the key to understanding the structure and properties of water, proteins. Here, quantum molecular dynamics simulations Hydrogen bonding is a fundamental element of chemical structure and reactivity. For an ion-dipole interaction to take effect, the ion and polar molecule in question must be in substantially closer proximity than is the case for hydrogen bonds. Water ionization becomes much less evident if the hydrogen bonds are just a few percent stronger but pure water contains considerably more H + ions if they are. Interactions can be studied simultaneously. 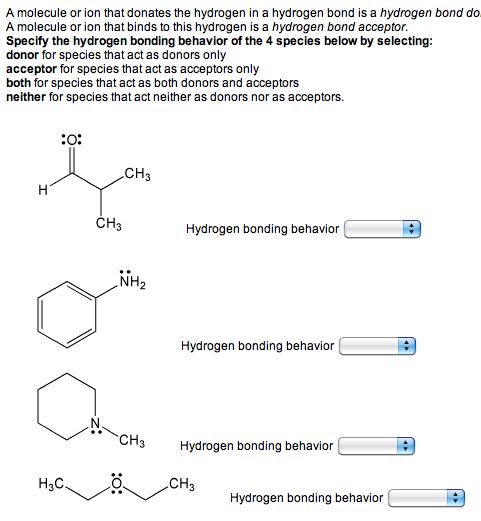
In this context, halide-dihydrate complexes are the smallest systems where the interplay between halide–water and water–water A hydrogen bond is an attraction between two atoms that already participate in other chemical bonds. The diaquahydrogen ion is of the asymmetric non-centred type. 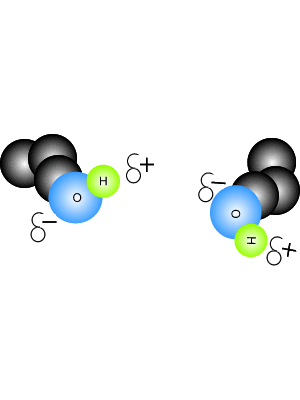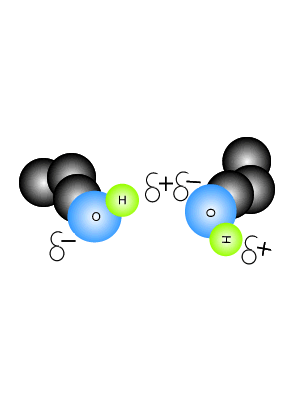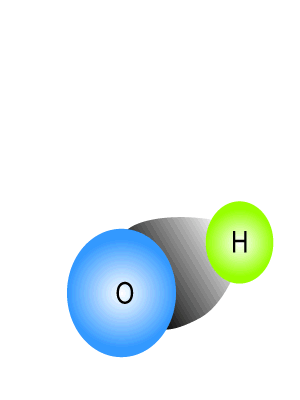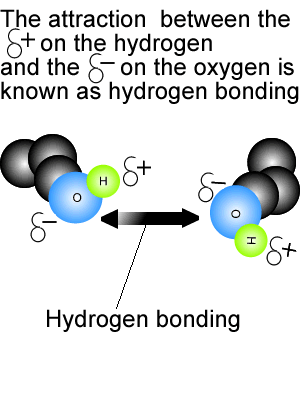
Small aqueous ionic clusters represent ideal systems to investigate the microscopic hydrogen-bonding structure and dynamics in ion hydration shells. The structure comprises H502+ ions, picrylsulphonate ions and water molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
